 Smart traders are beginning to take notice that the Awesome Penny Stocks play, Pristine Solutions (PRTN), doesn’t have the same “kool aid” flavor as past plays. With an obvious new writer for all the alerts disseminated, PRTN stock is still trading like an APS play, up 100% for some early entrants from Friday last week. The question now poised is whether the traders on the pump and dump will believe that FDA approval is within reach and able to push the share price to trade over the level that today’s emails are calling for.
Smart traders are beginning to take notice that the Awesome Penny Stocks play, Pristine Solutions (PRTN), doesn’t have the same “kool aid” flavor as past plays. With an obvious new writer for all the alerts disseminated, PRTN stock is still trading like an APS play, up 100% for some early entrants from Friday last week. The question now poised is whether the traders on the pump and dump will believe that FDA approval is within reach and able to push the share price to trade over the level that today’s emails are calling for.
![]() Our New Alert Is Set For Mid-September! Click HERE To Make Sure You Don’t Miss It!
Our New Alert Is Set For Mid-September! Click HERE To Make Sure You Don’t Miss It!
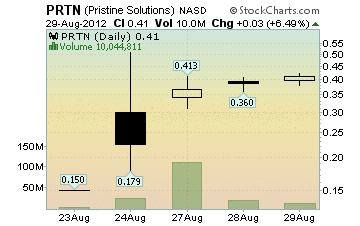 PRTN Stock Chart
PRTN Stock Chart
Market Cap: 171.38 Million
Close: 0.41, up 0.025 (6.49%)
Volume: 10,044,811
Dollar Volume: $4,104,619
High: 0.421
Low: 0.38
Trades: 1,466
Average Trade Size: 6,851
Authorized: 650 million
Issued and Outstanding: 418 million
![]() Looking for Hot Penny Stocks? Click here and get Alerts on the Best Penny Stocks FREE
Looking for Hot Penny Stocks? Click here and get Alerts on the Best Penny Stocks FREE
After yesterday’s close, Eaton Scientific Systems, Ltd., a wholly owned subsidiary of Pristine Solutions, announced that clinical trial veteran Dr. David L. Stark, D.C., Q.M.E., CCRA had been hired to develop and oversee FDA compliant trials of Tropine 3 for non-hormonal treatment of hot flashes in menopausal women. A nice dog and pony show for uneducated traders which have no clue as to what is involved in the FDA approval process.
How To Obtain FDA Approval
Drug companies continuously analyze thousands of compounds, seeking ones of therapeutic value. During the 6 to 7 years of preclinical testing, the manufacturer completes synthesis and purification of the drug and conducts limited animal testing. Of 5,000 compounds tested, approximately 5 will appear promising enough to induce the company to file an Investigational New Drug Application (IND). If the IND is approved by the FDA and by an Institutional Review Board, the manufacturer may begin the first phase of development.
The IND stage consists of 3 phases.
In phase I, clinical trials using healthy individuals are conducted to determine the drug’s basic properties and safety profile in humans. Typically the drug remains in this stage for 1 to 2 years. In phase II, efficacy trials begin as the drug is administered to volunteers of the target population. At the end of phase II, the manufacturer meets with FDA officials to discuss the development process, continued human testing, any concerns the FDA may have, and the protocols for phase III, which is usually the most extensive and most expensive part of drug development.
During the phases of the IND, the manufacturer can obtain accelerated development/review of the drug. Other accommodations for usage prior to approval include treatment IND and parallel tracking.
Once phase III is complete, the manufacturer files an NDA. Review of the NDA typically lasts 1 to 2 years, bringing total drug development and approval (that is, the IND and NDA stages) to approximately 9 years.
During the NDA stage, the FDA consults advisory committees made of experts to obtain a broader range of advice on drug safety, effectiveness, and labeling. Once approved, the drug may be marketed with FDA regulated labeling. The FDA also gathers safety information as the drug is used and adverse events are reported, and it will occasionally request changes in a labeling or will submit press releases as new contraindications arise. If adverse events appear to be systematic and serious, the FDA may withdraw a product from the market.
Over time there has been a clear tendency for FDA regulations and requirements to expand and multiply. In 1980, the typical drug underwent 30 clinical trials involving about 1,500 patients. By the mid-1990s, the typical drug had to undergo more than 60 clinical trials involving nearly 5,000 patients.
With that said, PRTN doesn’t stand a chance of seeing any FDA paperwork in the near-term other than a list of requirements they will need to achieve in order to even consider being a candidate. Harry Lappa and his North Springs Resources (NSRS) are more likely to find gold than Pristine Solutions’ (PRTN) Tropine 3 is to ever be FDA approved.
About PRTN Stock
Pristine Solutions, Inc., through its wholly-owned subsidiary company, Eaton Scientific Systems, Ltd., holds the intellectual property and global marketing rights to Tropine 3, a patent pending novel indication of an existing FDA approved drug for the non-hormonal treatment of hot flashes in pre-menopausal, pari-menopausal, and post-menopausal women. Insiders own shares of PRTN stock at a cost basis of $0.00025. (see here)
Bottom Line: The pump is surely on for this penny stock and should begin attracting attention if they continue to double and triple print transaction orders. Smart traders will be getting out of PRTN stock prior to the weekend and coming back in on the first dip after this first, uneventful, week of trading.
Here is your opportunity to subscribe to the Most Read Penny Stock Newsletter
If You Can’t Make Money With Us, You Shouldn’t Be Trading Penny Stocks
(We are 100% Anti-Spam and will never rent or sell your information)
ShareAUG

