![]() NYSE MKT listed Rexahn Pharmaceuticals, Inc. (RNN) saw a nice bounce on Friday after analysts at Burrill Institutional Research initiated coverage on the biopharmaceutical penny stock. Shares in the Oncology and CNS focused biotech have been bouncing around all year, dipping recently after closing a private placement worth $6.6 million. With the new coverage initiation by the Wall Street analysts at an “outperform” rating and a $1.00 price target given to RNN stock, a 212.5% upside, the cheap shares are worth watching as the Company appears to be adequately funded to start their Phase IIb clinical trials on their flagship drug, Archexin®, in the beginning of 2013.
NYSE MKT listed Rexahn Pharmaceuticals, Inc. (RNN) saw a nice bounce on Friday after analysts at Burrill Institutional Research initiated coverage on the biopharmaceutical penny stock. Shares in the Oncology and CNS focused biotech have been bouncing around all year, dipping recently after closing a private placement worth $6.6 million. With the new coverage initiation by the Wall Street analysts at an “outperform” rating and a $1.00 price target given to RNN stock, a 212.5% upside, the cheap shares are worth watching as the Company appears to be adequately funded to start their Phase IIb clinical trials on their flagship drug, Archexin®, in the beginning of 2013.
![]() Our Next Pick Is Coming Soon! Click HERE To Make Sure You Don’t Miss Any Of Our Breaking Alerts!
Our Next Pick Is Coming Soon! Click HERE To Make Sure You Don’t Miss Any Of Our Breaking Alerts!
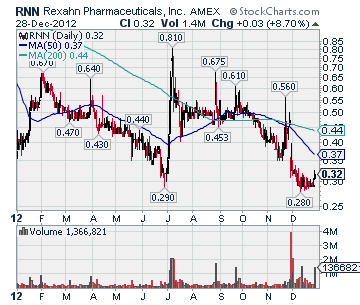 RNN Stock Chart
RNN Stock Chart
Market Cap: 30.27M
Close: 0.3175 ▲ 0.0254 (8.30%)
Volume: 1,366,821
Dollar Volume: $
High: 0.327
Low: 0.3058
Trades: 1,706
Average Trade Size: 801
52-Week:
High (Jul 9, 2012): 0.81
Low (Dec 18, 2012): 0.28
Moving Average:
50-Day: 0.37
200-Day: 0.44
![]() Looking for Hot Penny Stocks?
Looking for Hot Penny Stocks?
Click here and get Our Penny Stock Picks FREE
While many investors unfamiliar with Rexahn Pharmaceuticals may see the recent private placement, which was actually over allotted due to increased demand, as a cash grab for an cash poor biotech, the truth is they didn’t need it to stay afloat. No cash flows were provided from financing activities for the nine months ended September 30, 2012 and, as stated in their recent 10-Q, the Company had sufficient capital, $7,379,999, which was expected to be adequate to cover their cash flow requirements through September 30, 2013.
The private placement closing which took place at the beginning of the month, 19,130,435 shares and another 2,869,565 in the over allotment, raised almost as much capital as the Company had on hand at the end of Q3. Additionally, Teva Pharmaceutical Industries Ltd. completed a private offering of 2,083,333 shares of RNN stock for $750,000, $0.36 per share, to further develop RX-3117, an anti-cancer compound for which Rexahn owns the U.S. patent. So what is Rexahn planning to do with all this new cash?
The Company has developed two drugs which concentrate on Central Nervous System (CNS) disorders: Serdaxin® and Zoraxel™. While both are focused on varied release formulations of clavulanic acid, an ingredient present in antibiotics approved by the Food and Drug Administration (FDA), Serdaxin® which is designed for the treatment of depression and neurodegenerative disorders is set for the Phase IIb trial at little to no cost while Zoraxel™ which is designed for the treatment of erectile dysfunction would cost roughly $2,300,000 to continue forward with the Phase IIb trial. Neither of these two appear to be the next big thing.
Where Rexahn looks best prepared to be a biotech of the year candidate is in the forward testing of their flagship drug Archexin®, a 20 nucleotide single stranded DNA anti-sense molecule, which is a first-in-class inhibitor of the protein kinase Akt. Akt plays critical roles in cancer cell proliferation, survival, angiogenesis, metastasis, and drug resistance.
The Company’s Archexin received “orphan drug” designation from the FDA for five cancer indications: renal cell carcinoma, or RCC, glioblastoma, ovarian cancer, stomach cancer and pancreatic cancer. The FDA orphan drug program provides seven years of marketing exclusivity after approval and tax incentives for clinical research.
Five months ago, Rexahn announced top line results of their Phase IIa clinical trial which was designed to assess the safety and efficacy of Archexin in combination with gemcitabine. The study enrolled 31 subjects aged 18-65 with metastatic pancreatic cancer at nine centers in the United States and India with the primary endpoint being overall survival following four cycles of therapy with a six month follow-up. The Phase IIa study showed that treatment with Archexin in combination with gemcitabine provided a median survival rate of 9.1 months compared to the historical survival data of 5.65 months for standard single agent gemcitabine therapy according to data compiled from those evaluable patients with the most frequent reported adverse events being constipation, nausea, abdominal pain and pyrexia, regardless of relatedness.
Since Rexahn has indicated its intentions of advancing Archexin for a Phase IIb trial beginning in the first quarter of 2013, the long investors willing to sit on RNN stock for a good year plus could score big if the trials provide positive results. The short term investors may want to look at what happened in the beginning of 2012 when RNN stock ran up 80%. A PR issued that trial enrollment for Archexin is beginning may be all that’s needed to land this one in the “Win” column for the first double to start 2013.
About RNN Stock
Rexahn Pharmaceuticals, Inc. is a development stage biopharmaceutical company focusing on the development of novel cures for cancer to patients worldwide. The Company has yet to engage in any product sales until the Company receives approval from the FDA or equivalent foreign regulatory bodies to begin selling their pharmaceutical candidates. Click here to view the SEC filings for RNN, here to view their website.
Last 5 Trading Sessions:
| Date | Last | Change | % Change | Volume |
| 12/28/12 | 0.3175 | +0.0254 | +8.30% | 1,366,800 |
| 12/27/12 | 0.29 | unch | unch | 192,700 |
| 12/26/12 | 0.29 | unch | unch | 151,900 |
| 12/24/12 | 0.29 | unch | unch | 194,100 |
| 12/21/12 | 0.29 | unch | unch | 289,000 |
Bottom Line: Biotech penny stocks are always sexy, especially when you can get in for less than the insiders. With TEVA buying RNN at 0.36 and private placement purchasers buying at 0.33. The fact that the attached warrants in the private placement have an exercise price of 0.472 immediately puts RNN stock on the list of penny stocks for 2013.
Here is your opportunity to subscribe to the Most Read Penny Stock Newsletter
If You Can’t Make Money With Us, You Shouldn’t Be Trading Penny Stocks
(We are 100% Anti-Spam and will never rent or sell your information)
ShareDEC

