 The shares of Vitro Diagnostics Inc. (OTC: VODG) were the biggest gainers of the penny stocks on the OTC market in the last trading session with over $350k in volume. Vitro Diagnostics, which does business as Vitro Biopharma, reached as high as 0.268 during the trading session, a 239% gain, on the hottest of the hot penny stocks for the trading session. It was the biggest volume day ever for the penny stock, VODG, which had more trades executed than ECDC, DOMK, IPRC, WBSI, CBIS, NEOM, UCHC and even OMVE.
The shares of Vitro Diagnostics Inc. (OTC: VODG) were the biggest gainers of the penny stocks on the OTC market in the last trading session with over $350k in volume. Vitro Diagnostics, which does business as Vitro Biopharma, reached as high as 0.268 during the trading session, a 239% gain, on the hottest of the hot penny stocks for the trading session. It was the biggest volume day ever for the penny stock, VODG, which had more trades executed than ECDC, DOMK, IPRC, WBSI, CBIS, NEOM, UCHC and even OMVE.
Vitro Biopharma is actually quite unique. The stem cell company has been in business for over 20 years and been building revenues for the last 3 years. The most recent 10-K VODG filed showed a significant increase in net income from continuing operations.
![]() Looking for Hot Penny Stocks? Subscribe and get alerts on our next penny stock picks FREE.
Looking for Hot Penny Stocks? Subscribe and get alerts on our next penny stock picks FREE.
Top 10 Hot Penny Stocks
| Symbol | Close | Pct Chg | $ Volume | Volume | Trades |
|---|---|---|---|---|---|
| SEFE | 1.99 | +18.45% | 12,890,061 | 7,263,014 | 3,607 |
| SNPK | 0.66 | +19.57% | 9,151,874 | 14,664,532 | 3,442 |
| STVF | 0.68 | -18.27% | 2,069,458 | 3,120,045 | 1,163 |
| PLPE | 0.1065 | -27.05% | 660,630 | 5,420,368 | 655 |
| LQMT | 0.375 | -1.32% | 1,198,245 | 3,436,672 | 615 |
| LUXR | 1.22 | -2.40% | 1,216,559 | 1,029,193 | 575 |
| ICPA | 0.0265 | +12.77% | 871,312 | 30,487,811 | 543 |
| MSLP | 0.0195 | +13.37% | 668,527 | 34,868,374 | 514 |
| AAMRQ | 0.575 | -2.54% | 2,359,510 | 4,031,326 | 502 |
| VODG | 0.13 | +130.91 | 351,014 | 2,400,585 | 500 |
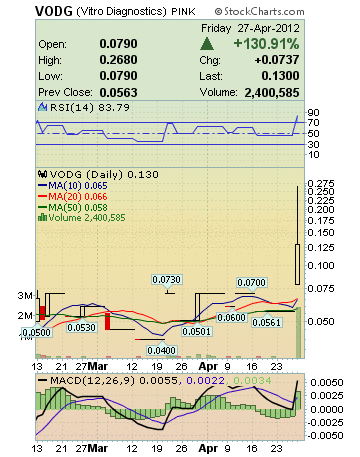 VODG Stock Snap Shot:
VODG Stock Snap Shot:
Market Cap: 2.45M
52-Week High
(Apr 27, 2012): 0.268
52-Week Low
(Mar 20, 2012): 0.04
Avg Vol (3 month): 48,295
Avg Vol (10 day): 300,438
Shares Outstanding: 18.87M
Float: 12.29M
Ytd Stock History:
Percent Change 116.67%
Moving Average 0.0545
Average Volume 39,764
Vitro Biopharma has some debt but the upside potential on VODG is encouraging. “Stem Cell” has been a hot buzzword in the penny stock investing and trading communities with companies like Advanced Cell Technology (OTC: ACTC) and Cord Blood America (OTC: CBAI). If these stem cell research companies like VODG can overcome the ethical and religious concerns with destroying human embryos, there is plenty of upside left to go on the penny stock.
About Vitro Diagnostics Inc.
Vitro was incorporated under the laws of the State of Nevada on February 3, 1986. From November 1990 to July 31, 2000, Vitro was engaged in the development, manufacture and distribution of purified human antigens and the development of therapeutic products and related technologies. In August 2000, Vitro sold the assets used in the manufacture and sale of purified antigens for diagnostic applications.
Since 2000, Vitro has developed its stem cell technology, expanded its patent portfolio and proprietary technology and cell lines for applications in stem cell research, cancer and diabetes. The VODG operations are currently focused on development, manufacturing and distribution of stem cell products and related tools for use in research and drug discovery. Vitro is presently expanding their stem cell product line to include products for use in clinical studies of the therapeutic benefits of stem cell transplantation.
Presently, Vitro develops, manufactures and markets products for use in research and related applications that do not require approval of the United States Food and Drug Administration prior to market introduction. VODG has previously developed products and product candidates for treatment of infertility.
Vitro launched its initial product line in 2009 and are implementing their early stage marketing program. The predominant focus of operations during the fiscal year ended October 31, 2011 was to continue commercialization of a series of products targeting worldwide markets in stem cell research, including products needed by most scientists engaged in stem cell research. These products are classified as Type I Medical Devices meaning that all usage is non-therapeutic.
During 2011, VODG expanded their product offerings considerably by adding new cell lines, especially media formulations to their 2011 Product Catalog. The VODG product offerings now include a specific type of human adult stem cell and certain derivatives of these cells, cell culture media for stem cell growth and differentiation into specific cell types together with various test kits for quantitative determination of stem cell quality, potency and response to toxic agents. The latter products are jointly manufactured by Vitro and their strategic partner, Hemogenix, Inc., a privately held stem cell firm located in Colorado Springs, CO, while all other products are developed and manufactured solely by Vitro.
During the quarter ended January 31, 2011, VODG completed an in-house study showing that its MSCGro stem cell culture media consistently improved mesenchymal stem cell proliferation, quality, potency and cellular recovery over leading competitors. Furthermore, MSCGro also exhibits real time stability that exceeds one year, while some competitors published recommended usage periods are two weeks to four weeks, thus resulting in substantial cost savings and expanded usage opportunities to MSCGro customers.
During 2011, Vitro also expanded its product line to include MSC-Gro Media suitable for use in clinical studies. Numerous investigations have shown the clinical value of mesenchymal stem cells in treatment of a variety of medical conditions. For example:
- diseases or injuries of the joints,
- cerebral palsy,
- autoimmune disorders, and
- enlarged heart resulting from prior heart attack, etc.
Thus, Vitro’s availability of clinical grade media opens opportunities for use of their MSC-Gro Media in clinical applications. VODG management views this component of their target market to be significant and plans to further expand its product line and regulatory approvals of its clinical grade MSC-Gro Media.
Vitro intends on pursuing FDA regulatory filings seeking appropriate approvals for clinical use of its media in the U.S.
To view the SEC filings for VODG, click here. For the current VODG stock quote, click here.
Bottom Line: Biotech stocks are tough to judge but when they run, you can’t catch them. It would have been nice to see VODG close on the HOD, but that can’t really be expected the first time that anyone outside the stem cell community had ever heard of them. Regardless, 130% gain for the trading session is nothing to complain about.
Add VODG to the list of penny stocks to watch for Monday. Remember not to chase the gaps in the event that the stock opens higher but watch its movements in the first 15 mins. VODG could be a one-hit wonder or another hidden gem that was lucky enough to have been found.
Click To Follow Us: 


ShareAPR

